The Multi-Ethnic Study of Atherosclerosis (MESA)
The Multi-Ethnic Study of Atherosclerosis (MESA) is a medical research study involving more than 6,000 men and women from six communities in the United States. MESA is sponsored by the National Heart Lung and Blood Institute of the National Institutes of Health. Participants in MESA are seen at clinics in the following universities:
Columbia University, New YorkJohns Hopkins University, Baltimore
Northwestern University, Chicago
UCLA, Los Angeles
University of Minnesota, Twin Cities
Wake Forest University, Winston Salem
About MESA
The Multi-Ethnic Study of Atherosclerosis (MESA) is a study of the characteristics of subclinical cardiovascular disease (disease detected non-invasively before it has produced clinical signs and symptoms) and the risk factors that predict progression to clinically overt cardiovascular disease or progression of the subclinical disease. MESA researchers study a diverse, population-based sample of 6,814 asymptomatic men and women aged 45-84. Approximately 38 percent of the recruited participants are white, 28 percent African-American, 22 percent Hispanic, and 12 percent Asian, predominantly of Chinese descent. Click here to read about MESA Overview and Protocol.
Invitation to New Investigators
We have found in many situations that a collaborative approach to research is
advantageous to affiliated and non-affiliated investigators alike.
Those who include MESA Investigators in their research are able to take
advantage of their considerable knowledge of the MESA dataset as well as
knowledge of research and analytic methodologies appropriate for the data.
In keeping with this collaborative approach, researchers interested in working
with MESA Investigators are welcome to submit a manuscript proposal or ancillary
study proposal directly to the study. Please feel free to review additional
materials related to establishing a collaborative relationship with MESA at the
following links (or send an e-mail to the Coordinating Center at
chsccweb@uw.edu).
https://www.mesa-nhlbi.org/Publications.aspx
https://www.mesa-nhlbi.org/ancillary.aspx
MESA Overview
The Multi-Ethnic Study of Atherosclerosis (MESA) is a study of the characteristics of subclinical cardiovascular disease (disease detected non-invasively before it has produced clinical signs and symptoms) and the risk factors that predict progression to clinically overt cardiovascular disease or progression of the subclinical disease. MESA researchers study a diverse, population-based sample of 6,814 asymptomatic men and women aged 45-84 from six field centers across the United States. Approximately 38 percent of the recruited participants are white, 28 percent African-American, 22 percent Hispanic, and 12 percent Asian, predominantly of Chinese descent. The first examination took place over two years, from July 2000-July 2002. It was followed by three examination periods that were 17-20 months in length, and a fifth exam April 2010 - January 2012. Participants are contacted every 9 to 12 months throughout the study to assess clinical morbidity and mortality. The final 18 months of the study are dedicated to close out and data analysis and publication.
Specific Analytic Opportunities
After careful review of the MESA publication record, the following list of analytic opportunities were identified as general topics where the data have not yet been adequately explored or published. There may be manuscripts in development, so it is strongly recommended that researchers with interest in collaborating on a research topic contact the point person directly (or the Coordinating Center at chsccweb@uw.edu) to initiate discussion and explore collaborative opportunities.
| Research Area | Point Person |
| Adaptive Immune System | Russ Tracy (Russell.Tracy@uvm.edu) |
| Atrial Fibrillation | Susan Heckbert (heckbert@u.washington.edu) |
| Bioelectric Impedance | Dick Kronmal (kronmal@uw.edu) |
| Coagulation | Mary Cushman (mary.cushman@med.uvm.edu) |
| Cognitive Function | Annette Fitzpatrick (fitzpal@u.washington.edu) |
| Diabetes | Alain Bertoni (abertoni@wakehealth.edu) |
| ECG Changes | Elsayed Soliman (esoliman@wakehealth.edu) |
| Genetic ancestry | Jasmin Divers (jdivers@wakehealth.edu) |
| Genetic Epi & Gene X Environ Int | Jerome Rotter (jrotter@labiomed.org) |
| Health Services Research | Chris Delaney (jacd@u.washington.edu) |
| Heart Failure | David Bluemke (bluemked@nih.gov) |
| Lipid Subclasses | Rachel Mackey (mackey@edc.pitt.edu) |
| Lipoprotein A [Lp(a)] | Mike Tsai (tsaix001@umn.edu) |
| Longitudinal data | Robyn McClelland (rmcclell@uw.edu) |
| Organ and System Fibrosis | Russ Tracy (Russell.Tracy@uvm.edu) |
| Policy and Prevention | Gregory L. Burke (gburke@wakehealth.edu) |
| Race/Ethnicity and Events | Wendy Post (wpost@jhmi.edu) |
| Stroke Clinical Events | Will Longstreth (wl@uw.edu) |
MESA Classic Components by Exam
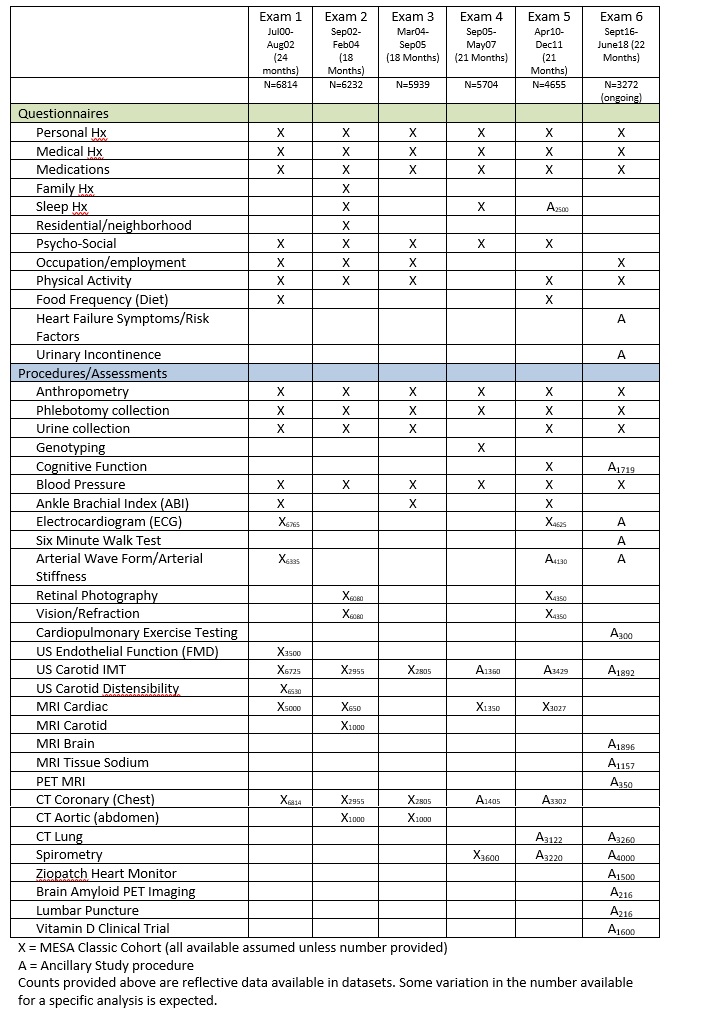
MESA III Timeline
MESA Calendar
Note: Click on the table below to get the full MESA Calendar image.

Organizational Chart

Key Scientific Personnel
| Site Type | Study Site | Key Personnel |
| Coordinating Center | University of Washington Coordinating Center |
Robyn McClelland, PhD (Co-PI) Richard A. Kronmal, PhD (Co-PI) Susan R. Heckbert, MD Bruce M. Psaty, MD Annette L. Fitzpatrick, PhD William T. Longstreth, MD Kayleen Williams, MPH Craig Johnson, MS |
| Field Centers | Columbia University Field Center |
Steven J. C. Shea, MD, MS (PI) Graham R. Barr, MD, Dr. P.H. Benjamin M. Smith, MD, MS, FRCPC Alan D. Guerci, MD Daniel Rabinowitz, PhD Alan R. Tall, MD |
| Johns Hopkins University Field Center |
Wendy S. Post, MD (PI) Erin D. Michos, MD, MHS Roger S. Blumenthal, MD Pamela Ouyang, MD A. Richey Sharrett, MD, DrPH Moyses Szklo, MD, MPH, Dr.Ph |
|
| Northwestern University Field Center |
Norrina B. Allen, PhD(PI) Kiang Liu, PhD (PI) Shah, Sanjiv J.MD Deepak K. Gupta, MD Thomas J. Wang, MD David Green, MD, PhD Philip Greenland, MD |
|
| UCLA Field Center |
Karol Watson, MD, PhD (PI) Preethi Srikanthan, MD, MS Alan M. Fogelman, MD Antoinette S. Gomes, MD Sameh Tadros |
|
| University of Minnesota Field Center |
Aaron R. Folsom, MD (PI) Lin Yee Chen, MD David R. Jacobs, Jr. PhD Michael Jerosch-Herold, PhD Pamela J. Schreiner, PhD James Pankow, PhD, MPH |
|
| Wake Forest University Field Center |
Gregory L. Burke, MD (PI) Alain G. Bertoni, MD Timothy Hughes, PhD Yongmei Liu, PhD Jingzhong Ding, PhD David M. Herrington, MD William G. Hundley, MD |
|
| Central Reading/Lab/Analysis Centers | University of Vermont Laboratory |
Russell P. Tracy, PhD (PI) Nels C. Olson, PhD Mary Cushman, MD Jon Peter Durda |
| Computed Tomography Reading Center
UCLA Medical Center Research and Education Institute |
Matthew J. Budoff, MD (PI) Robert C. Detrano, MD, PhD Chris Dailing |
|
| MRI Reading Center Johns Hopkins University |
Jo�o A. Lima, MD (PI) Bruce Wasserman, MD |
|
| Ultrasound Reading Center
New England Medical Center |
Joseph F. Polak, MD, MPH (PI) Daniel H. O'Leary, MD |
|
| ECG Reading Center
Wake Forest University |
Elsayed Z. Soliman, MD, MSc (PI) | |
| LA BioMed/Harbor UCLA |
Jerome I. Rotter, MD (PI) Xiuqing Guo, PhD Kent Taylor, PhD |
|
| University of Virginia Genetic Analysis Center |
Stephen Rich, PhD (PI) Josyf C. Mychaleckyj, MA, DPhil Michele M. Sale, PhD Ani Manichaikul, PhD |
|
| University of Minnesota Central Lipid Lab |
Michael Tsai, PhD (PI) John Eckfeldt, MD, PhD (Co-PI) |
|
| Program Office | National Heart, Lung, and Blood Institute |
Lorraine Silsbee, MHS Pothur R. Srinivas, PhD, MPH |
MESA Risk Score Calculator
An accurate estimate of 10-year CHD risk can be obtained using traditional risk factors and CAC.
The MESA risk score, which is available online on the MESA web site for easy use,
can be used to aid clinicians in the communication of risk to patients and when
determining risk-based treatment strategies.
This online calculator is most appropriate for patients in the 45-85 year age
range and in one of the following racial/ethnic groups: Caucasian, Chinese American, African American, or Hispanic.
To use the score you will need information on the following risk factors:
age, gender, race/ethnicity, diabetes (yes/no), current smoker (yes/no), total and HDL cholesterol,
use of lipid lowering medication (yes/no), systolic blood pressure (mmHg),
use of anti-hypertensive medication (yes/no), any family history
of heart attack in first degree relative (parent/sibling/child) (yes/no),
and a coronary artery calcium score (Agatston units).
Robyn L. McClelland, PhD; Neal W. Jorgensen, MS; Matthew Budoff, MD;
Michael J. Blaha, MD, MPH; Wendy S. Post, MD, MS; Richard A. Kronmal, PhD;
Diane E. Bild, MD, MPH; Steven Shea, MD, MS; Kiang Liu, PhD; Karol E. Watson, MD, PhD;
Aaron R. Folsom, MD; Amit Khera, MD; Colby Ayers, MS; Amir-Abbas Mahabadi, MD;
Nils Lehmann, PhD; Karl-Heinz Jockel, PhD Susanne Moebus, PhD; J. Jeffrey Carr, MD, MS;
Raimund Erbel, MD, PhD; Gregory L. Burke, MD, MS
10-Year Coronary Heart Disease Risk Prediction Using Coronary Artery Calcium and
Traditional Risk Factors: Derivation in the MESA (Multi-Ethnic Study of Atherosclerosis)
With Validation in the HNR (Heinz Nixdorf Recall) Study and the DHS (Dallas Heart Study).
J Am Coll Cardiol. 2015 Oct 13;66(15):1643-53.
MESA Risk Score Calculator please click 'Start' button below:
| Start Risk Score Calculator |
CAC Score Reference Values
Coronary Artery Calcium (CAC) Score Reference Values web tool will provide the
estimated probability of non-zero calcium, and the 25th, 50th, 75th, and 90th
percentiles of the calcium score distribution for a particular age, gender and
race. Additionally, if an observed calcium score is entered the program will
provide the estimated percentile for this particular score. These reference
values are based on participants in the MESA study who were free of clinical
cardiovascular disease and treated diabetes at baseline. These participants
were between 45-84 years of age, and identified themselves as White,
African-American, Hispanic, or Chinese. The current tool is thus applicable
only for these four race/ethnicity categories and within this age range.
At this time, the risk associated with a particular calcium score is unknown.
Thus, the information in this tool cannot necessarily be used to conclude that
a patient is "high risk", but can indicate whether they have a high calcium
score relative to others with the same age, gender, and race/ethnicity.
McClelland RL, Chung H, Detrano R, Post W, Kronmal RA.
Distribution of coronary artery calcium by race, gender, and age: results from
the Multi-Ethnic Study of Atherosclerosis (MESA).
Circulation. 2006;113(1):30-37.
To use CAC Score Reference Values web tool please click 'Start' button below:
| Start CAC Reference Values |
Arterial Age Calculator
Arterial age provides a convenient transformation of coronary artery calcium
(CAC) from Agatston units to age units, to a scale more easily appreciated by
both patients and treating physicians. The arterial age for a participant is
the age at which the estimated CHD risk (modeled as a function of age) is the
same as that for the observed CAC score. Arterial age is then the
risk-equivalent of coronary artery calcium. This measure can be considered a
more easily understandable version of the CAC score (e.g. you are 55 years old,
but your arteries are more consistent with an arterial age of 65 years).
This tool will calculate an estimated arterial age (and 95% confidence
interval) given a CAC score input by the user. Optionally, one can also provide
the observed age, gender, total cholesterol, HDL cholesterol, smoking status,
systolic blood pressure and use of anti-hypertensive medications and obtain two
versions of estimated 10-year CHD risk based on the Framingham (NCEP) point
based equations: one using original age, and the other using estimated arterial
age. This does not apply to diabetics.
Robyn
L. McClelland, PhD, Khurram Nasir, MD, MPH, Matthew Budoff, MD, Roger S.
Blumenthal, MD, and Richard A. Kronmal, PhD.
Arterial Age as a Function of Coronary Artery Calcium (from the Multi-Ethnic
Study of Atherosclerosis [MESA]), Am J Cardiol. 2009 January 1; 103(1): 59-63
To use Arterial Age Calculator please click the button below:
| Calculate Arterial Age |
MESA Publications
An Invitation to Outside Investigators
We have found in many situations that a collaborative approach to research is advantageous
to affiliated and non-affiliated investigators alike. Those who include MESA Investigators in
their research are able to take advantage of their considerable knowledge of the MESA dataset
as well as knowledge of research and analytic methodologies appropriate for the data.
In keeping with this collaborative approach, researchers interested in working with MESA
Investigators are welcome to submit a manuscript proposal or ancillary study proposal directly
to the study. Please feel free to review additional materials related to establishing a
collaborative relationship with MESA at the following links (or send an e-mail
to the Coordinating Center at chsccweb@uw.edu).
https://www.mesa-nhlbi.org/Publications.aspx
https://www.mesa-nhlbi.org/ancillary.aspx
MESA Ancillary Studies
Overview
Definition of an ancillary study:
1) A project that collects new data in MESA, whether directly
from participants or from previously collected samples, images,
or other sources (e.g., medical records).
2) A project that analyzes existing MESA data as part of a new
external funding application, for which additional MESA Coordinating
Center (CC) services will be requested beyond downloading of data already
available on the MESA website (e.g., analysis by a CC Statistician or
preparation of a unique dataset). Note: as of November, 2010, analysis-only
grants involving no such additional CC services require the submission of a
Manuscript Proposal form, but not an Ancillary Study Proposal Form. Submit
the Manuscript Proposal form online and see MESA Publications submission
details at: https://www.mesa-nhlbi.org/Publications.aspx.
To check the availability of online data, please contact your MESA Sponsor.
Philosophy: MESA investigators are encouraged to consider ancillary studies and to involve other investigators, within and outside of MESA, in this process.